Semantic search
| Term | Abbreviation | Description |
|---|---|---|
| PH calibration buffers | pH calibration buffers are prepared to obtain two or more defined pH values for calibration of pH electrodes and pH indicator dyes. | |
| POS calibration - dynamic | Calibration of the sensor response time. See also POS calibration - static. | |
| POS calibration - static | F5 | Two-point calibration of the polarographic oxygen sensor, comprising Air calibration and Zero calibration. See also POS calibration - dynamic. |
| Pathway control ratio | FCR | Substrate control ratios are flux control ratios FCR, at a constant mitochondrial coupling-control state. Whereas there are only three well-defined coupling-control states of mitochondrial respiration, L, P, E (LEAK respiration, OXPHOS, Electron transfer pathway), numerous Electron-transfer-pathway states are possible. Careful selection of the reference state, Jref, is required, for which some guidelines may be provided without the possibility to formulate general rules. FCR are best defined by taking Jref as the maximum flux (e.g. NSE), such that flux in various other respiratory states, Ji, is smaller or equal to Jref. However, this is not generally possible with FCR. For instance, the N/S pathway control ratio (at constant coupling-control state) may be larger or smaller than 1.0, depending on the mitochondrial source and various mitochondrial injuries. The S-pathway control state may be selected preferentially as Jref, if mitochondria with variable N-linked injuries are studied. In contrast, the reference state, Z, is strictly defined for flux control efficiency. |
| Photodecomposition | PD | Photodecomposition or photodegradation is the process of decay of organic material induced by increasing light intensity. Under aerobic conditions, the enhancement of photodecomposition by light intensity can be quantified by oxygen consumption in a controlled light regime. |
| Physiological pathway-control state | See Electron-transfer-pathway state. | |
| Polyether ether ketone | PEEK | Polyether ether ketone (PEEK) is a semicrystalline organic polymer thermoplastic, which is chemically very resistant, with excellent mechanical properties. PEEK is compatible with ultra-high vacuum applications, and its resistance against oxygen diffusion make it an ideal material for high-resolution respirometry (POS insulation; coating of stirrer bars; stoppers for closing the O2k-Chamber). |
| Polyvinylidene fluoride | PVDF | Polyvinylidene fluoride (PVDF) is a pure thermoplastic fluoropolymer, which is chemically very resistant, with excellent mechanical properties. It is used generally in applications requiring the highest purity, strength, and resistance to solvents, acids, bases and heat (Wikipedia). PVDF is resistant against oxygen diffusion which makes it an ideal material for high-resolution respirometry (coating of stirrer bars; stoppers for closing the O2k-Chamber). |
| Power O2k-FluoRespirometer | Power O2k-FluoRespirometer - optional configuration as additional system for increasing output combined with the O2k-FluoRespirometer (O2k-Series H). The Power O2k-FluoRespirometer includes the TIP2k and the O2k-sV-Module, and supports all add-on O2k-Modules of the Oroboros O2k. It can be added to an existing Oroboros O2k of any O2k-Series. This application does not require an additional ISS-Integrated Suction System and O2k-Titration Set. Furthermore, the OroboPOS-Mounting Tool of the OroboPOS Service Tools can be used from the available O2k and is not included. | |
| Power O2k-Respirometer | The Power O2k-Respirometer is an economical option for using additional O2k-Units to increase output in high-resolution respirometry. | |
| Proton leak | Flux of protons driven by the protonmotive force across the inner mt-membrane, bypassing the ATP synthase and thus contributing to LEAK respiration. Proton leak-flux depends non-linearly (non-ohmic) on the protonmotive force. Compare: Proton slip. | |
| Proton pump | Mitochondrial proton pumps are large enzyme complexes (CI, CIII, CIV, CV) spanning the inner mt-membrane, partially encoded by mtDNA. CI, CIII and CIV are proton pumps that drive protons against the electrochemical protonmotive force, driven by electron transfer from reduced substrates to oxygen. In contrast, ATP synthase (also known as CIV) is a proton pump that utilizes the energy of proton flow along the protonmotive force to drive phosphorylation of ADP to ATP. | |
| Proton slip | Proton slip is a property of the proton pumps (Complexes CI, CIII, and CIV) when the proton slips back to the matrix side within the proton pumping process. Slip is different from the proton leak, which depends on Δp and is a property of the inner mt-membrane (including the boundaries between membrane-spanning proteins and the lipid phase). Slip is an uncoupling process that depends mainly on flux and contributes to a reduction in the biochemical coupling efficiency of ATP production and oxygen consumption. Together with proton leak and cation cycling, proton slip is compensated for by LEAK respiration or LEAK oxygen flux, L. Compare: Proton leak. | |
| Q calibration - DatLab | Q calibration | |
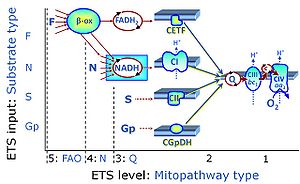
| Q-junction |
The Q-junction is a junction for convergent electron flow in the Electron transfer pathway (ET-pathway) from type N substrates and mt-matrix dehydrogenases through Complex I (CI), from type F substrates and FA oxidation through electron-transferring flavoprotein Complex (CETF), from succinate (S) through Complex II (CII), from glycerophosphate (Gp) through glycerophosphate dehydrogenase Complex (CGpDH), from choline through choline dehydrogenase, from dihydro-orotate through dihydro-orotate dehydrogenase, and other enzyme Complexes into the Q-cycle (ubiquinol/ubiquinone), and further downstream to Complex III (CIII) and Complex IV (CIV). The concept of the Q-junction, with the N-junction and F-junction upstream, provides the rationale for defining Electron-transfer-pathway states and categories of SUIT protocols. | |
| ROUTINE respiration | R | |
| Respiratory acceptor control ratio | RCR | The respiratory acceptor control ratio (RCR) is defined as State 3/State 4 [1]. If State 3 is measured at saturating [ADP], RCR is the inverse of the OXPHOS control ratio, L/P (when State 3 is equivalent to the OXPHOS state, P). RCR is directly but non-linearly related to the P-L control efficiency, jP-L = 1-L/P, with boundaries from 0.0 to 1.0. In contrast, RCR ranges from 1.0 to infinity, which needs to be considered when performing statistical analyses. In living cells, the term RCR has been used for the ratio State 3u/State 4o, i.e. for the inverse L/E ratio [2,3]. Then for conceptual and statistical reasons, RCR should be replaced by the E-L coupling efficiency, 1-L/E [4]. |
| Respiratory chain | RC | The mitochondrial respiratory chain (RC) consists of enzyme complexes arranged to form a metabolic system of convergent pathways for oxidative phosphorylation. In a general sense, the RC includes (1) the electron transfer pathway (ET-pathway), with transporters for the exchange of reduced substrates across the inner mitochondrial membrane, enzymes in the matrix space (particularly dehydrogenases of the tricarboxylic acid cycle), inner membrane-bound electron transfer complexes, and (2) the inner membrane-bound enzymes of the phosphorylation system. |
| Respiratory complexes | CI, CII, CIII, CIV, CETF, CGpDH, .. | Respiratory Complexes are membrane-bound enzymes consisting of several subunits which are involved in energy transduction of the respiratory system. » MiPNet article |
| Respiratory state | Respiratory states of mitochondrial preparations and living cells are defined in the current literature in many ways and with a diversity of terms. Mitochondrial respiratory states must be defined in terms of both, the coupling-control state and the electron-transfer-pathway state. | |
| Respirometry | Respirometry is the quantitative measurement of respiration. Respiration is therefore a combustion, a very slow one to be precise (Lavoisier and Laplace 1783). Thus the basic idea of using calorimetry to explore the sources and dynamics of heat changes were present in the origins of bioenergetics (Gnaiger 1983). Respirometry provides an indirect calorimetric approach to the measurement of metabolic heat changes, by measuring oxygen uptake (and carbon dioxide production and nitrogen excretion in the form of ammonia, urea, or uric acid) and converting the oxygen consumed into an enthalpy change, using the oxycaloric equivalent. Liebig (1842) showed that the substrate of oxidative respiration was protein, carbohydrates, and fat. The sum of these chemical changes of materials under the influence of living cells is known as metabolism (Lusk 1928). The amount (volume STP) of carbon dioxide expired to the amount (volume STP) of oxygen inspired simultaneously is the respiratory quotient, which is 1.0 for the combustion of carbohydrate, but less for lipid and protein. Voit (1901) summarized early respirometric studies carried out by the Munich school on patients and healthy controls, concluding that the metabolism in the body was not proportional to the combustibility of the substances outside the body, but that protein, which burns with difficulty outside, metabolizes with the greatest ease, then carbohydrates, while fats, which readily burns outside, is the most difficultly combustible in the organism. Extending these conclusions on the sources of metabolic heat changes, the corresponding dynamics or respiratory control was summarized (Lusk 1928): The absorption of oxygen does not cause metabolism, but rather the amount of the metabolism determines the amount of oxygen to be absorbed. .. metabolism regulates the respiration. | |
| STPD | STPD | At standard temperature and pressure dry (STPD: 0 °C = 273.15 K and 1 atm = 101.325 kPa = 760 mmHg), the molar volume of an ideal gas, Vm, and Vm,O2 is 22.414 and 22.392 L∙mol-1, respectively. Rounded to three decimal places, both values yield the conversion factor of 0.744 from units used in spiroergometry (VO2max [mL O2·min-1]) to SI units [µmol O2·s-1]. For comparison at normal temperature and pressure dry (NTPD: 20 °C), Vm,O2 is 24.038 L∙mol-1. Note that the SI standard pressure is 100 kPa, which corresponds to the standard molar volume of an ideal gas of 22.711 L∙mol-1 and 22.689 L∙mol-1 for O2. |
| SUIT | SUIT | SUIT is the abbreviation for Substrate-Uncoupler-Inhibitor Titration. SUIT protocols are used with mt-preparations to study respiratory control in a sequence of coupling and pathway control states induced by multiple titrations within a single experimental assay. These studies use biological samples economically to gain maximum information with a minimum amount of cells or tissue. |
| SUIT-001 | RP1 | 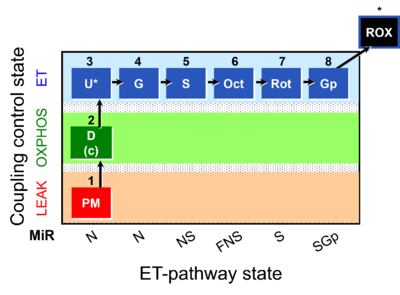 |
| SUIT-001 O2 ce-pce D003 | RP1 ce-pce |  |
| SUIT-001 O2 ce-pce D004 | RP1 ce-pce blood | 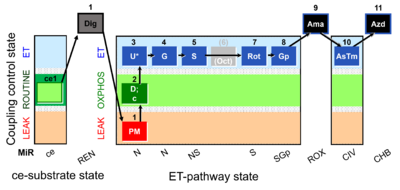 |
| SUIT-001 O2 mt D001 | RP1 mt | 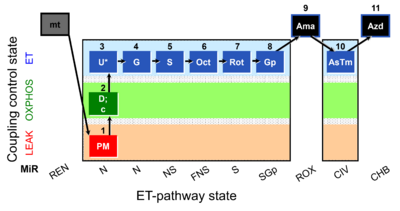 |
| SUIT-001 O2 pfi D002 | RP1 pfi | 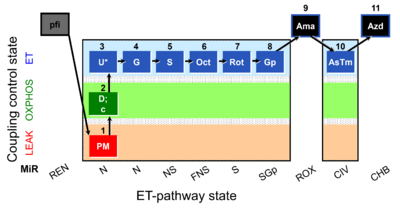 |
| SUIT-002 | RP2 | 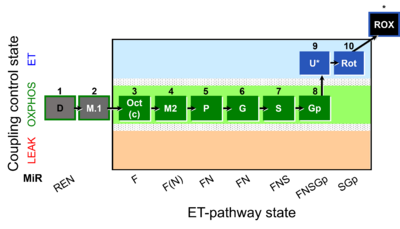 |
| SUIT-002 O2 ce-pce D007 | RP2 ce-pce | 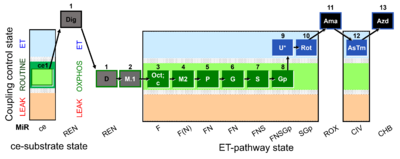 |
| SUIT-002 O2 ce-pce D007a | RP2 ce-pce blood |  |
| SUIT-002 O2 mt D005 | RP2 mt | 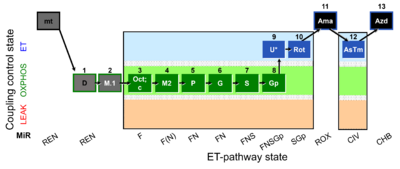 |
| SUIT-002 O2 pfi D006 | RP2 pfi | 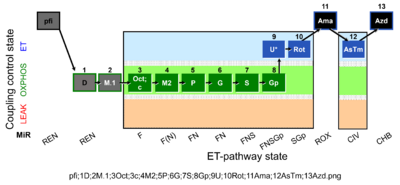 |
| SUIT-003 | CCP-ce | 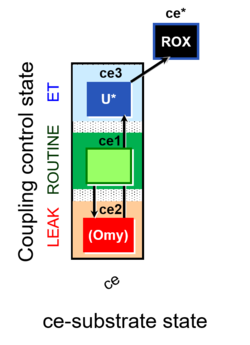 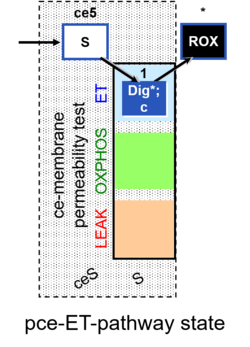 |
| SUIT-003 AmR ce D058 | AmR effect on ce | 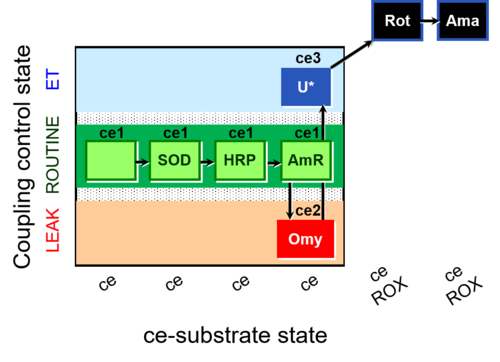 |
| SUIT-003 AmR ce D059 | AmR effect on ce - control | 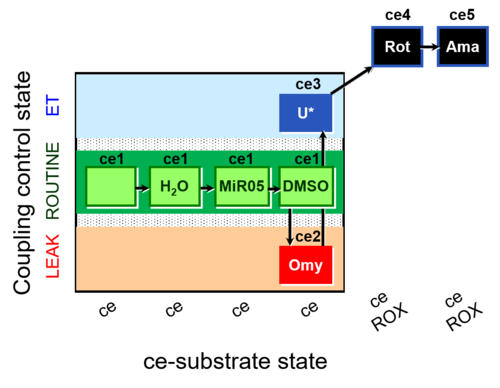 |
| SUIT-003 Ce1;ce1P;ce3U;ce4Glc;ce5M;ce6Rot;ce7S;1Dig;1c;2Ama;3AsTm;4Azd | cePMGlc,S | 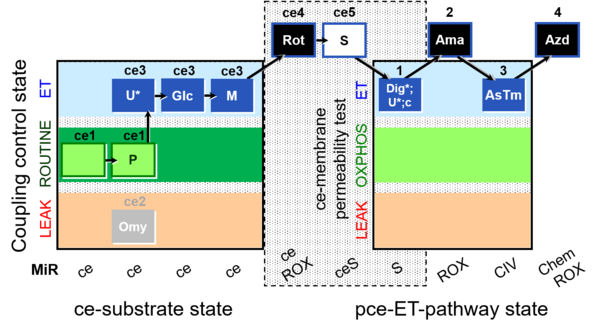 |
| SUIT-003 Ce1;ce2U- | ce | 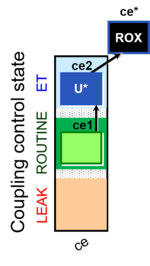 |
| SUIT-003 Ce1;ce3U- | ce |  |
| SUIT-003 O2 ce D009 | CCP-ce short | 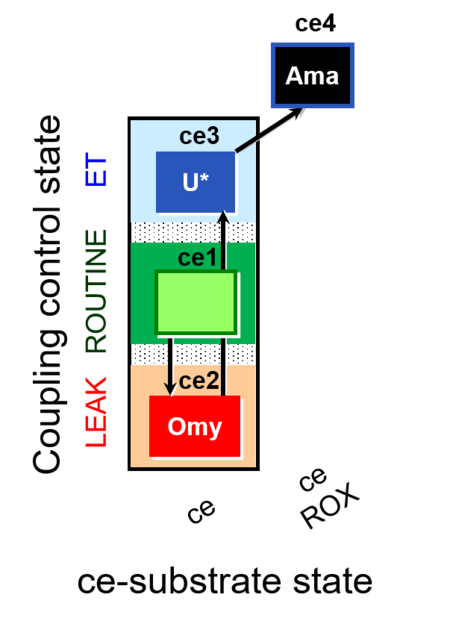 |
| SUIT-003 O2 ce D012 | CCP-ce(P) | 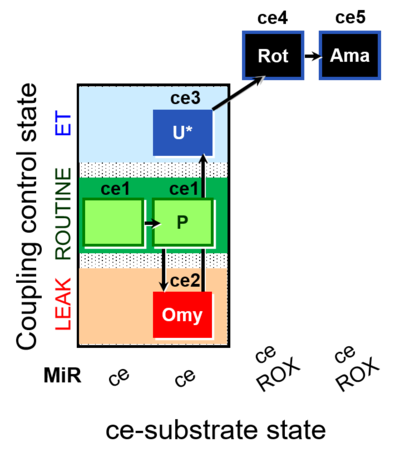 |
| SUIT-003 O2 ce D028 | CCP-ce S permeability test | 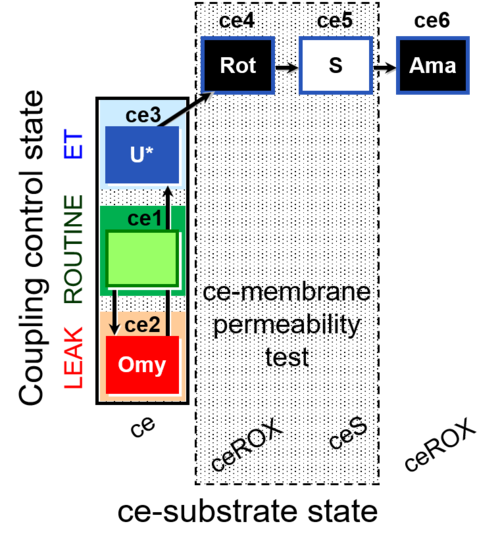 |
| SUIT-003 O2 ce D037 | CCP-ce Crabtree_R |  |
| SUIT-003 O2 ce D038 | CCP-ce Crabtree_E |  |
| SUIT-003 O2 ce D039 | CCP-ce microalgae | 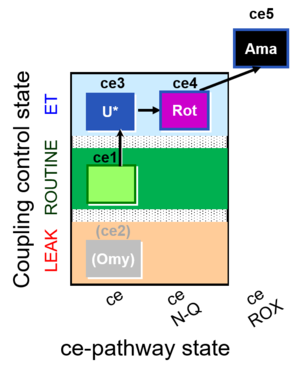 |
| SUIT-003 O2 ce D050 | CCP-ce Snv | 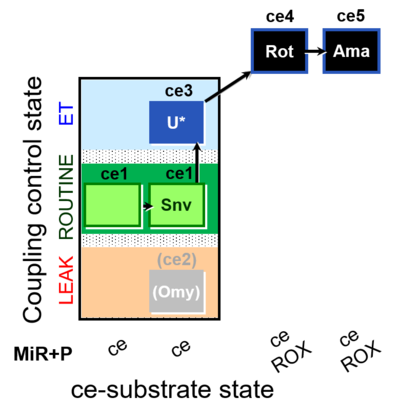 |
| SUIT-003 O2 ce D060 | CCP-ce Snv,Mnanv | 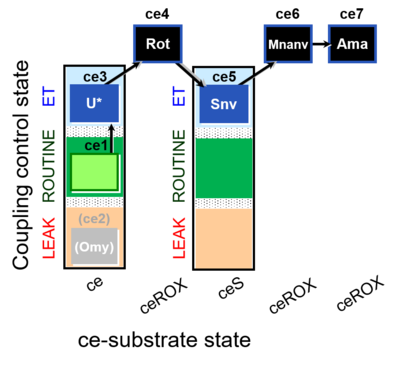 |
| SUIT-003 O2 ce D061 | CCP-ce Snv,Mnanv - control | 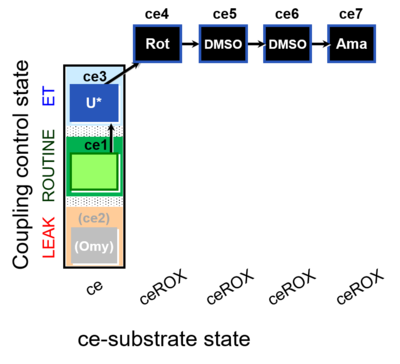 |
| SUIT-003 O2 ce D062 | CCP-ce Snv - control | 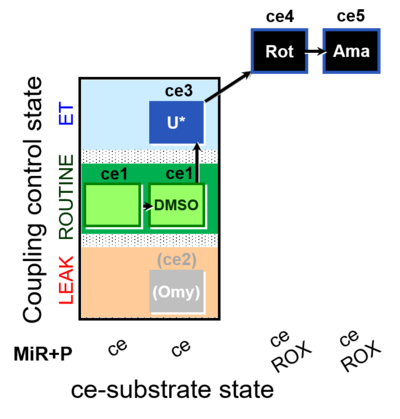 |
| SUIT-003 O2 ce-pce D013 | CCVP-Glc,M | 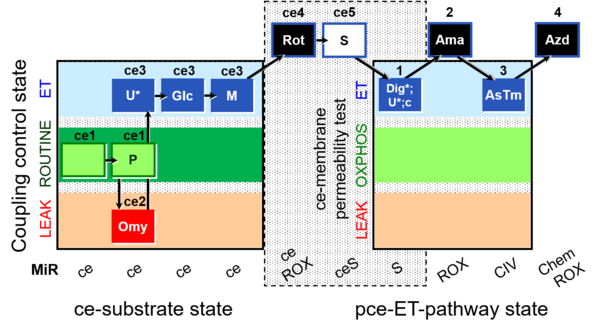 |
| SUIT-003 O2 ce-pce D018 | CCVP-Glc | 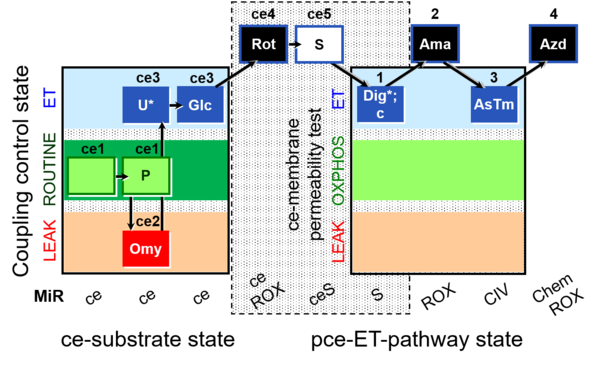 |
| SUIT-003 O2 ce-pce D020 | CCVP | 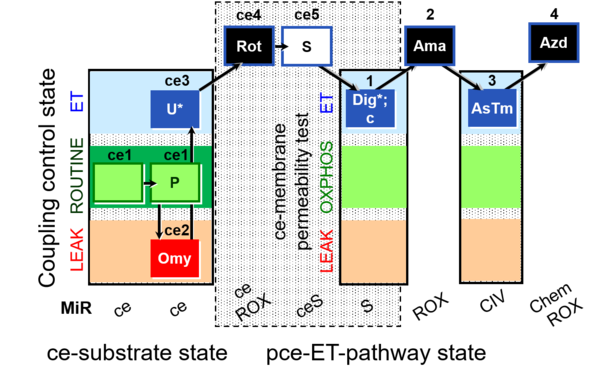 |
| SUIT-003 pH ce D067 | CCP-Crabtree with glycolysis inhibition | 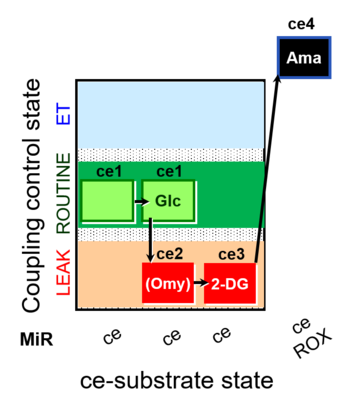 |
| SUIT-004 | RP1-short |  |
| SUIT-004 O2 pfi D010 | RP1-short pfi | 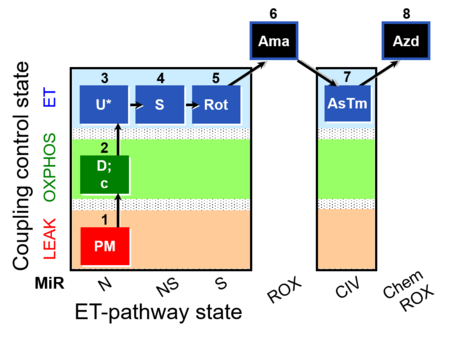 |
| SUIT-005 | RP2-short | 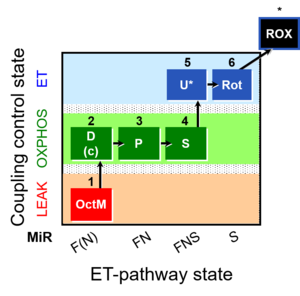 |
| SUIT-005 O2 pfi D011 | RP2-short pfi | 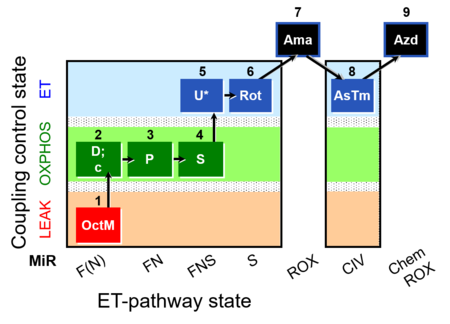 |
| SUIT-006 | CCP-mtprep | 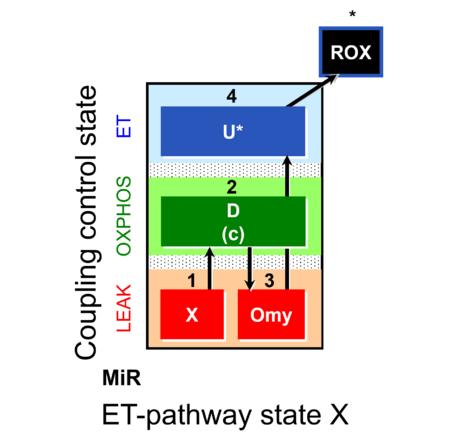 |
| SUIT-006 O2 ce-pce D029 | CCP ce-pce PM | 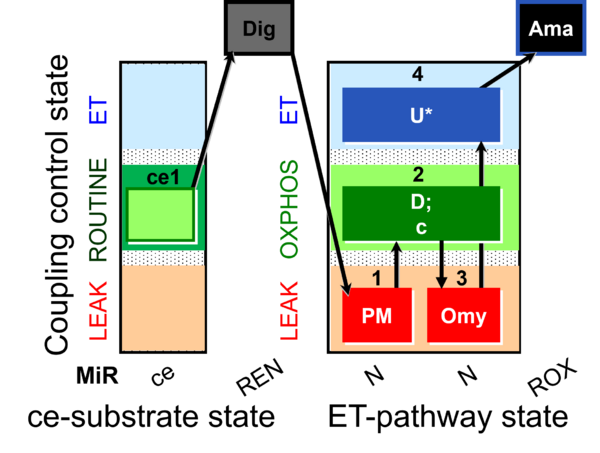 |
| SUIT-006 O2 mt D022 | CCP mt S(Rot) | 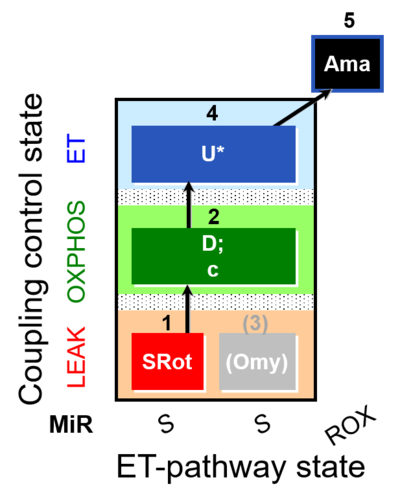 |
| SUIT-006 O2 mt D047 | CCP mt PM | 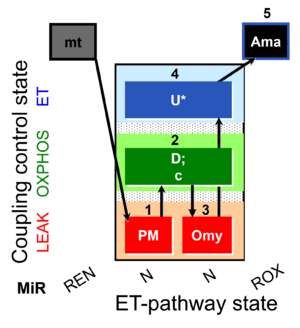 |
| SUIT-006 Q ce-pce D073 | CCP ce-pce S(Rot) | 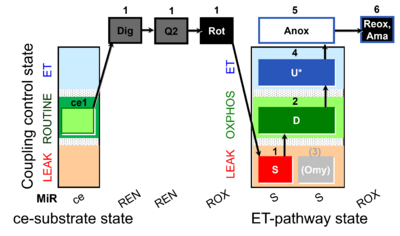 |
| SUIT-006 Q mt D071 | CCP mt S(Rot) | 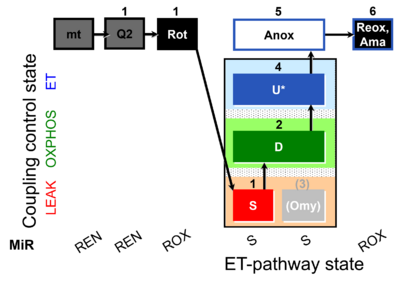 |
| SUIT-007 | Glutamate anaplerosis |  |
| SUIT-007 O2 ce-pce D030 | Glutamate anaplerotic pathway |  |
| SUIT-008 | PM+G+S_OXPHOS+Rot_ET | 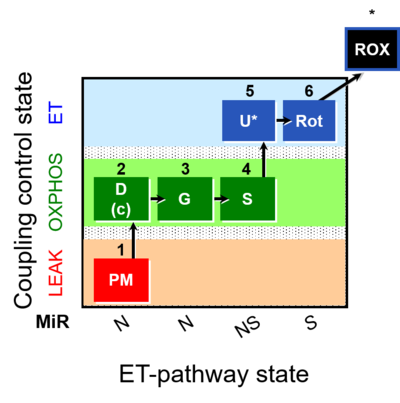 |
| SUIT-008 O2 ce-pce D025 | Q-junction ce-pce | 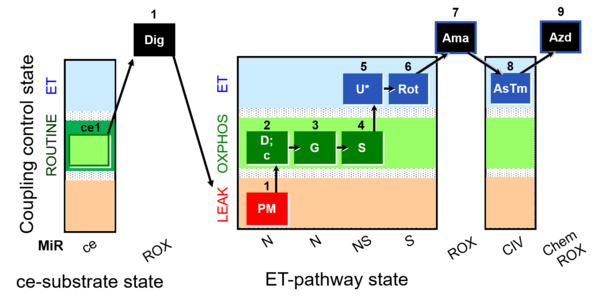 |
| SUIT-008 O2 mt D026 | Q-junction mtprep | 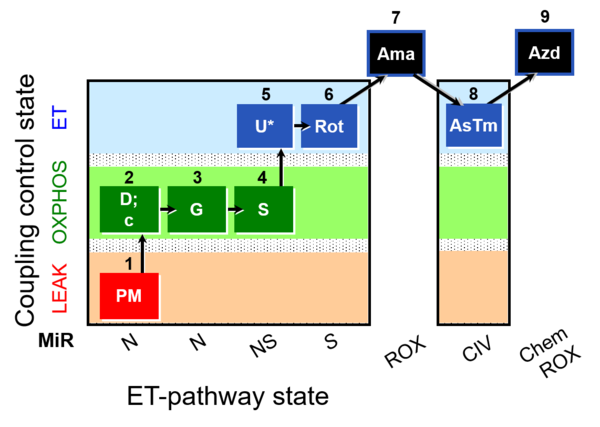 |
| SUIT-008 O2 pce D25 | NS(PGM) | 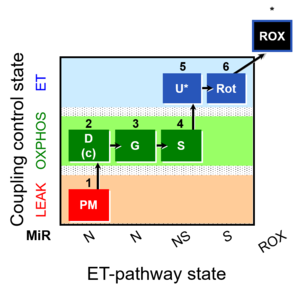 |
| SUIT-008 O2 pfi D014 | 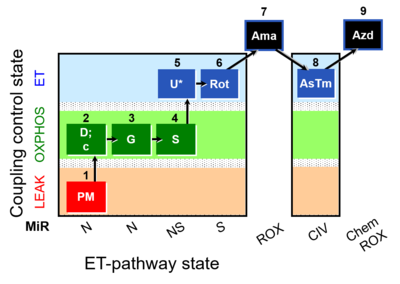 | |
| SUIT-009 O2 ce-pce D016 |  | |
| SUIT-009 O2 mt D015 | 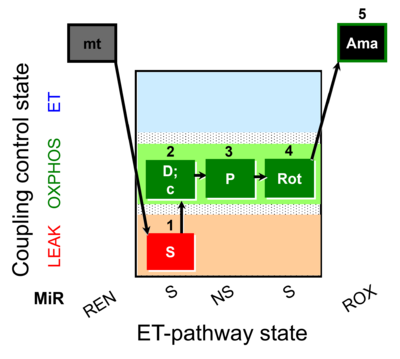 | |
| SUIT-010 | Digitonin test | 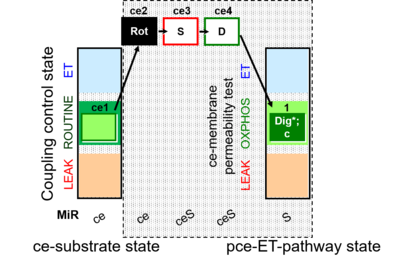 |
| SUIT-010 O2 ce-pce D008 | Dig titration-pce |  |
| SUIT-011 | GM+S_OXPHOS+Rot_ET | 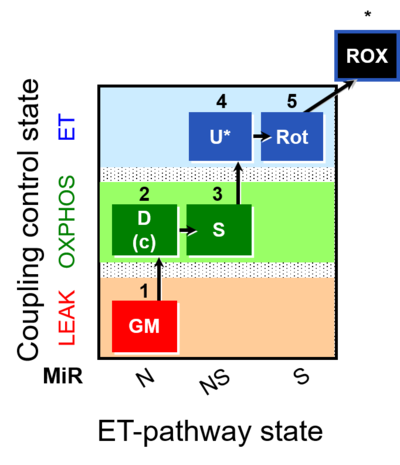 |
| SUIT-011 O2 pfi D024 | NS physiological maximum capapcity in fibres | 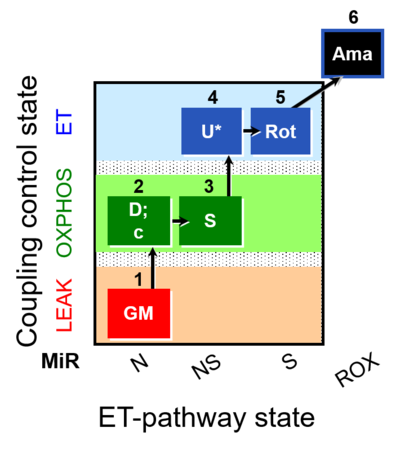 |
| SUIT-012 | PM+G_OXPHOS | 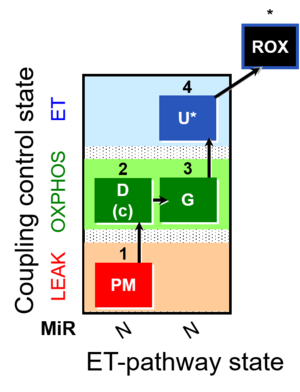 |
| SUIT-012 O2 ce-pce D052 | N(PGM) | 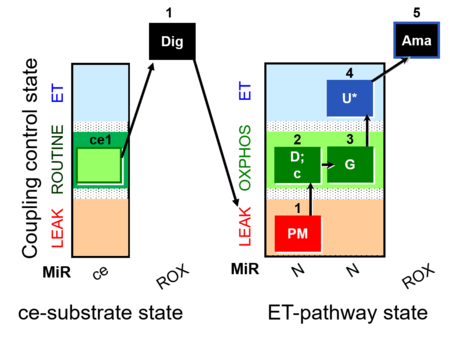 |
| SUIT-012 O2 mt D027 | N CCP mtprep | 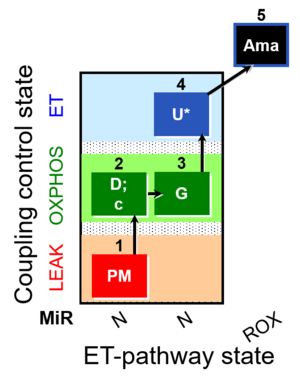 |
| SUIT-013 AmR ce D023 | O2 dependence of H2O2 production ce | 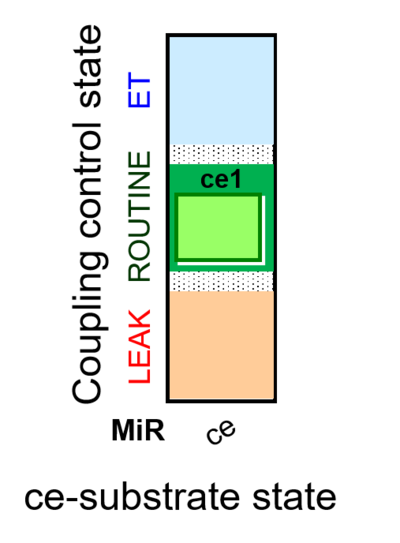 |
| SUIT-014 | GM+P+S_OXPHOS+Rot_ET |  |
| SUIT-014 O2 pfi D042 | NS(PGM) |  |
| SUIT-015 | F+G+P+S_OXPHOS+Rot_ET |  |
| SUIT-015 O2 pti D043 | FNS(Oct,PGM) |  |
| SUIT-016 | F+G+S+Rot_OXPHOS+Omy | 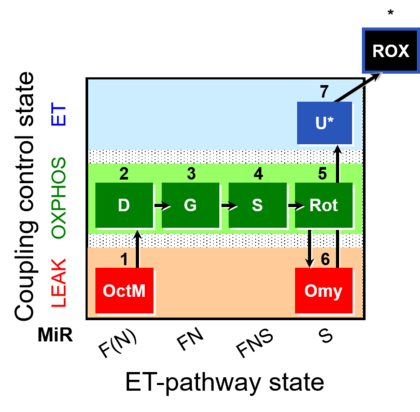 |
| SUIT-016 O2 pfi D044 | FNS(Oct,GM) | 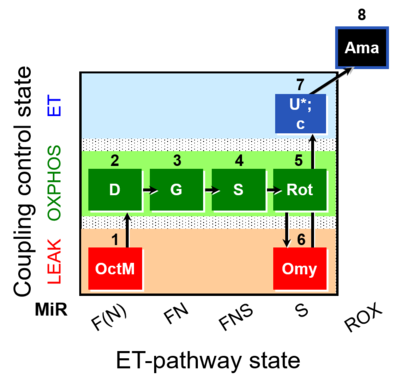 |
| SUIT-017 | F+G+S_OXPHOS+Rot_ET | 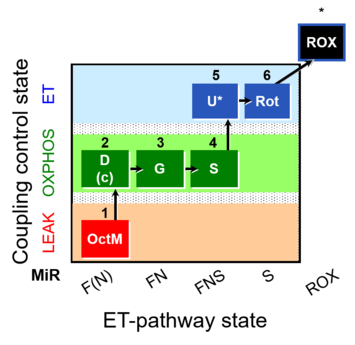 |
| SUIT-017 O2 mt D046 | FNS(Oct,GM) | 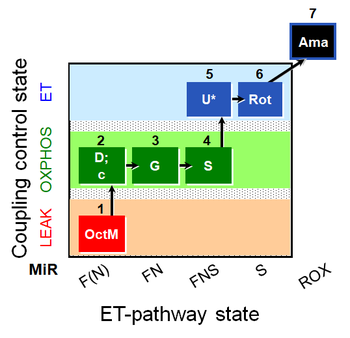 |
| SUIT-017 O2 pfi D049 | FNS(Oct,GM) |  |
| SUIT-018 O2 mt D054 | 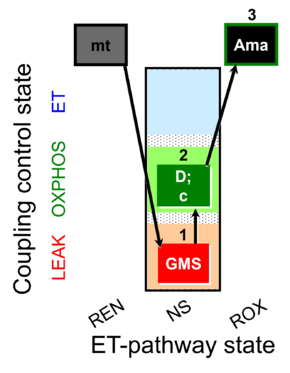 |