From Bioblast
MitoFit-Reference sample
- The project MitoFit highlights the benefits of mitochondrial fitness.
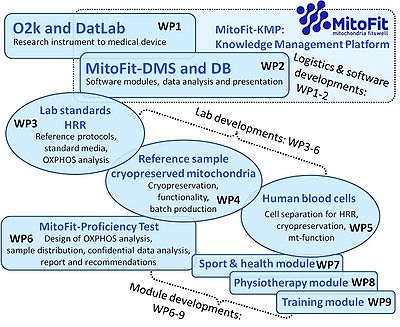
WP4
- Reference sample: cryopreserved mitochondria - development for functional control assays
Abstract
- A laboratory quality control system requires proficiency tests (MitoFit proficiency test) based on repeated assays (QCS) of a reference sample (WP4). Appropriate reference samples are not available for mitochondrial respiratory research. According to the EUROPEAN COMMISSION, a reference sample is “a sample of a batch of starting material, packaging material or finished product which is stored for the purpose of being analysed should the need arise during the shelf life of the batch concerned. ... Reference samples are for the purpose of analysis and, therefore, should be conveniently available to a laboratory with validated methodology.” In the present context, this implies that in order to serve as a respirometric refrence sample, the reference bulk material must be produced in sufficiently large quantities and homogeneity for splitting into subsamples. Vial to vial variability must be negligible relative to the error of diagnostic assays, to avoid sample variability as a confounding factor in the detection of systemic errors of measurement. Reference samples should retain preserved properties during transport and storage for prolonged periods of time, such that a single batch will last at least for one year. The reference sample should approximate the nature of biomedical and patient specimens with respect to experimental SUIT protocols to be applied.
Cryopreservation
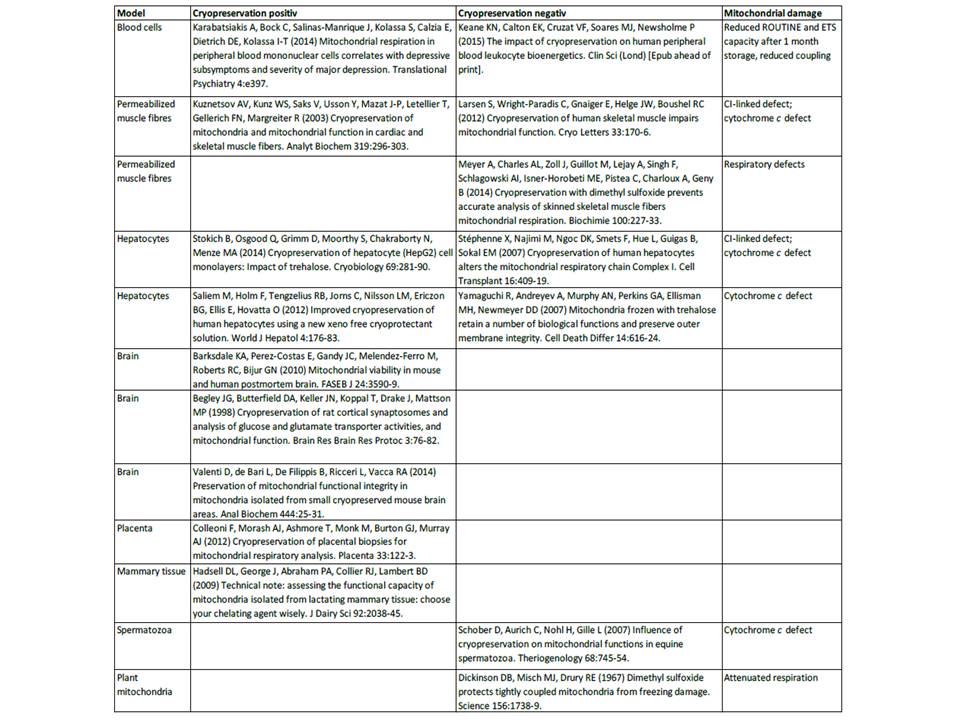
- The current lack of a reference sample offering sufficient stability of respiratory parameters (intact mitochondrial membranes, preserved mitochondrial respiratory capacity, coupled oxidative phosphorylation) during storage has so far prevented implementation of proficiency testing. Application of stabilizers such as dimethylsulfoxide and polyethylene glycol yields controversial results on preserving mitochondrial function (Tab. 1). Mitochondrial respiratory competence has been reported in mononuclear human blood cells directly after storage at -80 °C (Karabatsiakis et al 2014). Since cultured cells are routinely stored at -80 °C and cultured after thawing, it appears most promising to optimize conditions for cryopreservation of respiratory functions in a selected mammalian cell culture.
- Table 1
Progress and next steps
- » Doerrier 2016 Abstract MitoFit Science Camp 2016: Development of a SUIT reference protocol for OXPHOS analysis by high-resolution respirometry.
- » Lamberti 2016 Abstract MitoFit Science Camp 2016: Development of a reference sample for HRR.
Links and references
- » Emons H, Fajgelj A, van der Veen AMH, Watters R (2006) New definitions on reference materials. Accred Qual Assur 10:576-8.